
Used pharmaceutical freeze dryer





Different Parameters, Different Price.
Our staff will give you the most suitable quote.
- Year : 2014
- dimension: 2500 mm x 1253 mm
- Max.bowl speed: LYO-3
- weight: 4000 kg
Introduction
This is Tofflon’s LYO-3 pharmaceutical vacuum freeze dryer, designed for drug processing scenarios (e.g., freeze-drying of APIs, biological preparations). It has an effective shelf area of 3.24 m² and a 60 kg ice capture capacity, complying with pharmaceutical production standards, and is suitable for small-to-medium batch pharmaceutical freeze-drying processes.
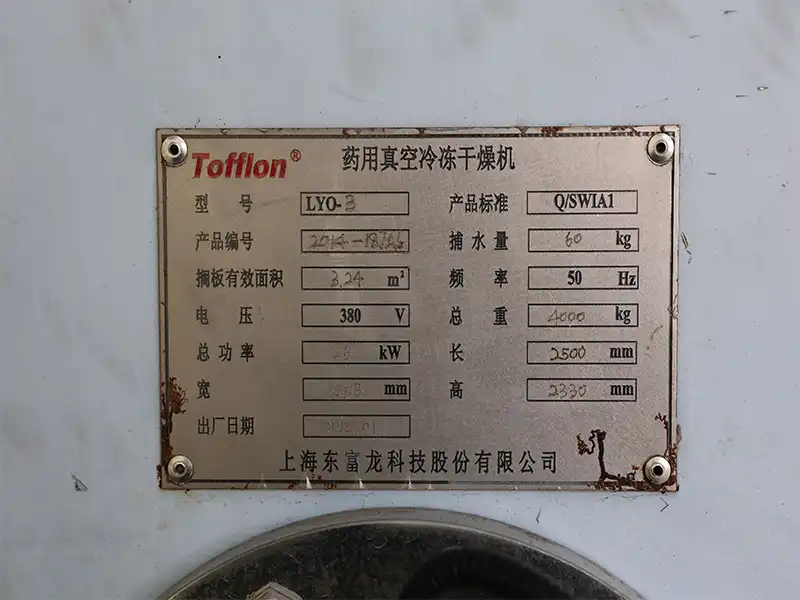

Specification
Item | Details |
Equipment Name | Pharmaceutical Vacuum Freeze Dryer |
Brand | Tofflon |
Model | LYO-3 |
Product Standard | Q/SWIA1 |
Product No. | 2014-127A6 |
Effective Shelf Area | 3.24 m² |
Condenser Ice Capacity | 60 kg |
Voltage | 380 V |
Frequency | 50 Hz |
Total Power | 26 kW |
Gross Weight | 4000 kg |
Length | 2500 mm |
Width | 1253 mm |
CONDENSER
Item | Details |
Equipment Name | Shell and Tube Condenser |
Brand | Tofflon |
Product Model | WSZ098 |
Product No. | EL114768 |
Vessel Category | Category I |
Manufacturing Date | December 5, 2014 |
Vessel Net Weight | 65 kg |
Heat Transfer Area | 4.2 m² |
Baffle Spacing | / mm |
Product Standard | NB/T47012-2010 |
Manufacturing License Level | D1, D2 |
Manufacturing License No. | TS2231009-2017 |
Manufacturer | Shanghai Tofflon Pressure Vessel Co., Ltd. |
Equipment Code | 913102000250141432 |
Tube Side Parameters | — |
Design Pressure | 0.6 MPa |
Hydrostatic Test Pressure | 0.7 MPa |
Maximum Allowable Working Pressure | 0.6 MPa |
Design Temperature | 55°C |
Working Medium | Water |
Main Material | T2 |
Product Features
- The elegant appearance of medical machinery, compact integrated design, and portable transportation;
- Equipped with automatic defrosting for the condenser and automatic sealing for vials, without steam sterilization disinfection function;
- Flexible process formula design, users can customize 12 steps during the freeze-drying stage, and make real-time adjustments to the freeze-drying process formula during the experiment, meeting the process development requirements for various products;
- Fully automatic operation, with a user-friendly human-machine interface design, simple and easy to understand;
- Adopting a stable SCADA (Data Acquisition and Supervisory Control) standard modular system, enabling data acquisition, analysis, recording, and automation of the freeze-drying process;
- Equipped with three control modes: manual, automatic, and semi-automatic. It also includes functions such as electronic signature, electronic records, audit tracking, and permission management, meeting the FDA 21 CFR PART 11 requirements.





