
Used pharmaceutical freeze dryer





Different Parameters, Different Price.
Our staff will give you the most suitable quote.
- Manufacturing Date : 2011.12
- Size: 2810 mm x 1050 mm x 1980 mm
- brand: Tofflon
- Compressor Weight : 2500 kg
Introduction
The vacuum freeze-drying machine (referred to as the freeze-drying machine) is a device for carrying out the freeze-drying process. Freeze-drying is a process where a substance or solution is frozen and dried at low temperatures. By freezing the water-containing substance or solution in a sealed container into ice (in a solid state), and then heating it in a vacuum environment, the water in the water-containing substance or solution is removed.

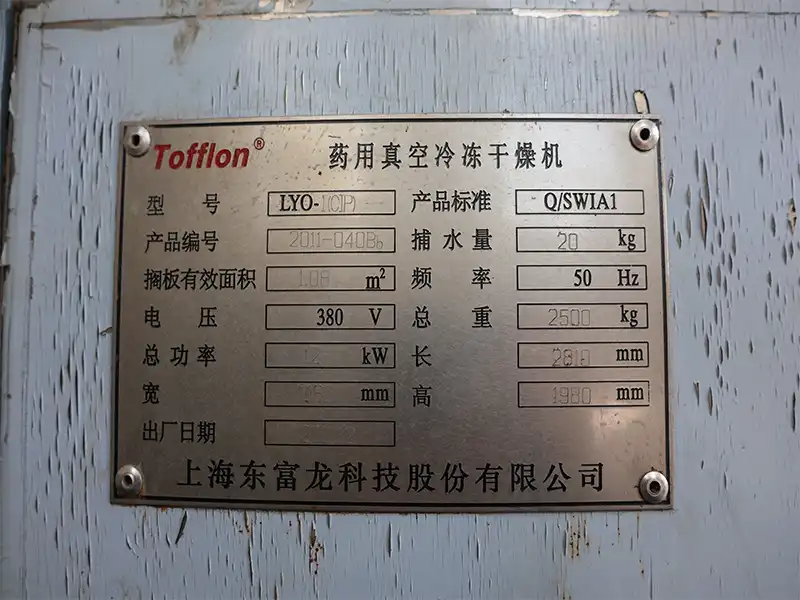
Specification
Item | Specification |
Equipment Name | Pharmaceutical Vacuum Freeze Dryer |
Brand | Tofflon |
Model | LYO-1(CIP) |
Product Standard | Q/SWIA1 |
Product Serial Number | 2011-0408b |
Effective Shelf Area | 1.08 m² |
Water Catching Capacity | 20 kg |
Voltage | 380 V |
Frequency | 50 Hz |
Total Power | 12 kW |
Total Weight | 2500 kg |
Length | 2810 mm |
Width | 1050 mm |
Height | 1980 mm |
Product Features
- The elegant appearance of medical machinery, compact integrated design, and portable transportation;
- Equipped with automatic defrosting for the condenser and automatic sealing for vials, without steam sterilization disinfection function;
- Flexible process formula design, users can customize 12 steps during the freeze-drying stage, and make real-time adjustments to the freeze-drying process formula during the experiment, meeting the process development requirements for various products;
- Fully automatic operation, with a user-friendly human-machine interface design, simple and easy to understand;
- Adopting a stable SCADA (Data Acquisition and Supervisory Control) standard modular system, enabling data acquisition, analysis, recording, and automation of the freeze-drying process;
- Equipped with three control modes: manual, automatic, and semi-automatic. It also includes functions such as electronic signature, electronic records, audit tracking, and permission management, meeting the FDA 21 CFR PART 11 requirements.





